PIB Headquarters
કોવિડ-19 અંગે PIBનું દૈનિક બુલેટિન
Posted On:
07 DEC 2021 6:08PM by PIB Ahmedabad


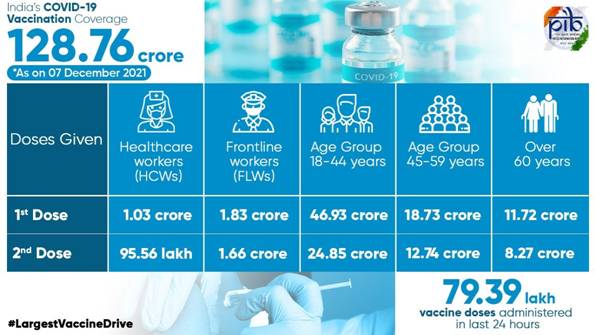
- રાષ્ટ્રવ્યાપી રસીકરણ અભિયાન અંતર્ગત અત્યાર સુધી 128.76 કરોડ લોકોને રસી આપવામાં આવી
- ભારતમાં સક્રિય કેસનું ભારણ તાજેતરમાં 95,014, 554 દિવસમાં સૌથી ઓછું
- સક્રિય કેસ કુલ કેસનાં 1% કરતા ઓછા છે, હાલમાં 0.27% છે, માર્ચ 2020 પછી સૌથી ઓછા
- સાજા થવાનો દર હાલમાં 98.36% નોંધાયો
- છેલ્લા 24 કલાકમાં 10,004 દર્દીઓ સાજા થયા, કુલ વધીને 3,40,79,612 દર્દીઓ સાજા થયા
- છેલ્લા 24 કલાકમાં ભારતમાં 6,822 નવા કેસ નોંધાયા, 558 દિવસમાં સૌથી ઓછા
- દૈનિક પોઝિટિવીટી દર 0.63% પહોંચ્યો, છેલ્લા 64 દિવસથી 2% કરતા ઓછો
- સાપ્તાહિક પોઝિટિવીટી દર છેલ્લા 23 દિવસથી 1% કરતા ઓછો થઈ ગયો છે, હાલમાં 0.78% છે
- કુલ 64.94 કરોડ પરીક્ષણો કરવામાં આવ્યા
#Unite2FightCorona
#IndiaFightsCorona
PRESS INFORMATION BUREAU
MINISTRY OF INFORMATION & BROADCASTING
GOVERNMENT OF INDIA
*****

કોવિડ-19 અપડેટ
વિગત: https://pib.gov.in/PressReleasePage.aspx?PRID=1778689
રાજ્યો/કેન્દ્રશાસિત પ્રદેશોમાં કોવિડ-19 રસીની ઉપલબ્ધતા અંગે અપડેટ
વિગત: https://pib.gov.in/PressReleasePage.aspx?PRID=1778680
વિવિધ પ્રકારના રોગચાળાને નિયંત્રિત કરવા માટે ડોકટરો અને પેરા-મેડિકલ સ્ટાફને વિશેષ તાલીમ
વિગત: https://pib.gov.in/PressReleasePage.aspx?PRID=1778830
કોવિડ-19 મહામારીના કોઈપણ પુનરુત્થાનના જોખમને ઘટાડવા માટે લેવામાં આવેલા પગલાં
વિગત: https://pib.gov.in/PressReleasePage.aspx?PRID=1778832
કોવિડ-19 રસીની નિકાસ
વિગત: https://pib.gov.in/PressReleasePage.aspx?PRID=1778837
Vaccination Program for Children and Under-18 Population
National Expert Group on Vaccine Administration for Covid (NEGVAC) and National Technical Advisory Group on Immunization (NTAGI) are deliberating and considering scientific evidences related to vaccination of beneficiaries aged less than 18 years.
Government of India has been supplying Covid-19 vaccines free of cost to the States/UTs for administration to prioritized beneficiaries as recommended by NEGVAC.
ZyCoV-D vaccine manufactured by M/s Cadila Healthcare has received the approval for Restricted Use in Emergency Setting by the National Regulator i.e., Drug Controller General of India (DCGI) for the age group of 12 years and above based on the interim clinical data of Phase II &Phase III clinical trial conducted in the country.
Following COVID-19 vaccines are under clinical trials in the country for age-group of less than 18 years:
- M/s Bharat Biotech is conducting Phase II/III clinical trial of COVAXIN on Healthy Volunteers aged 2 to 18 years & firm has submitted interim safety & immunogenicity data to the National Regulator.
- M/s Serum institute of India is conducting Phase II/III clinical trial of Nanoparticle Vaccine (Liquid) (COVOVAX) in 920 subjects of >2 to 17 years age group.
- M/s Biological E Ltd., is conducting Phase II/III clinical trial of RBD of SARS-CoV-2 gene in 624 subjects of ≥5 to <18years age group.
- M/s Johnson & Johnson Pvt. Ltd., is conducting Phase II/III clinical trial of Ad.26COV.2S vaccine in age group of 12-17 years (Global Clinical trial wherein India is one of the clinical trial sites).
The approval of aforementioned COVID-19 vaccines is dependent on the successful completion of clinical trials and submission of requisite data to the National Regulator i.e., Drug Controller General of India as per the requirements of New Drugs and Clinical Trials Rules, 2019 under Drugs and Cosmetics Act, 1940.
Inclusion of Diseases like Covid-19 and Dengue under AB-PMJAY
Treatment of diseases like COVID-19 and dengue is included under Ayushman Bharat –Pradhan Mantri Jan Arogya Yojana (AB-PMJAY). Details of specific Health Benefit Packages for COVID-19 and dengue are at Annexure-I. State-wise details of COVID-19 testing and treatment under AB-PMJAY are at Annexure-II.
Annexure-I
Details of specific Health Benefit Packages for COVID-19 under AB-PMJAY:
|
Sl. No.
|
Package Name
|
Procedure Name
|
|
1
|
Laboratory Tests for COVID-19 Infection (PCR) (Reimbursement level for this package will be as per the ICMR guidelines, issued from time to time)
|
Laboratory Tests for COVID-19 Infection (PCR) (Reimbursement level for this package will be as per the ICMR guidelines, issued from time to time)
|
|
2
|
Laboratory Tests for COVID-19 Infection (PCR) (Reimbursement level for this package will be as per the ICMR guidelines, issued from time to time)
|
Laboratory Tests for COVID-19 Infection (PCR) (Reimbursement level for this package will be as per the ICMR guidelines, issued from time to time)
|
|
3
|
Treatment of COVID-19 Infection
|
Treatment of COVID-19 Infection
|
Details of specific Health Benefit Packages for dengue under AB-PMJAY:
|
Sl. No.
|
Package Name
|
Procedure Name
|
|
1
|
Dengue Fever
|
Dengue Fever
|
|
2
|
Dengue Fever
|
Dengue Hemorrhagic Fever
|
|
3
|
Dengue Fever
|
Dengue Shock Syndrome
|
Annexure-II
COVID-19 Pre-authorization raised under AB-PMJAY:
|
State/UT
|
Testing Count
|
Treatment Count
|
|
Andaman and Nicobar Islands
|
32
|
7
|
|
Andhra Pradesh
|
|
2,00,945
|
|
Assam
|
332
|
1,028
|
|
Bihar
|
145
|
22
|
|
Chandigarh
|
3
|
7
|
|
Chhattisgarh
|
1,049
|
43,964
|
|
DNH and DD
|
19
|
4
|
|
Goa
|
|
1
|
|
Gujarat
|
11,355
|
|
|
Haryana
|
14,817
|
719
|
|
Himachal Pradesh
|
12
|
52
|
|
Jammu and Kashmir
|
4
|
729
|
|
Jharkhand
|
30
|
1,495
|
|
Karnataka
|
|
1,82,070
|
|
Kerala
|
18,728
|
1,33,591
|
|
Madhya Pradesh
|
7,891
|
18,352
|
|
Maharashtra
|
1,35,904
|
1,82,991
|
|
Manipur
|
1
|
732
|
|
Meghalaya
|
7,589
|
3,932
|
|
Mizoram
|
|
416
|
|
Nagaland
|
-
|
12
|
|
Puducherry
|
20
|
349
|
|
Punjab
|
13
|
|
|
Rajasthan
|
|
23,761
|
|
Sikkim
|
-
|
32
|
|
Tamil Nadu*
|
18,50,134
|
31,076
|
|
Tripura
|
1
|
54
|
|
Uttar Pradesh
|
1,571
|
1,421
|
|
Uttarakhand
|
2,042
|
2,801
|
|
Total
|
20,51,692
|
8,29,826
|
Note:
* Treatment data is as of 25th October 2021; Testing data is as of 1st week of June 2021
SD/GP/JD
સોશિયલ મીડિયા પર અમને ફોલો કરો :  @PIBAhmedabad
@PIBAhmedabad  /pibahmedabad1964
/pibahmedabad1964  /pibahmedabad
/pibahmedabad  pibahmedabad1964[at]gmail[dot]com
pibahmedabad1964[at]gmail[dot]com
(Release ID: 1779051)
Visitor Counter : 128